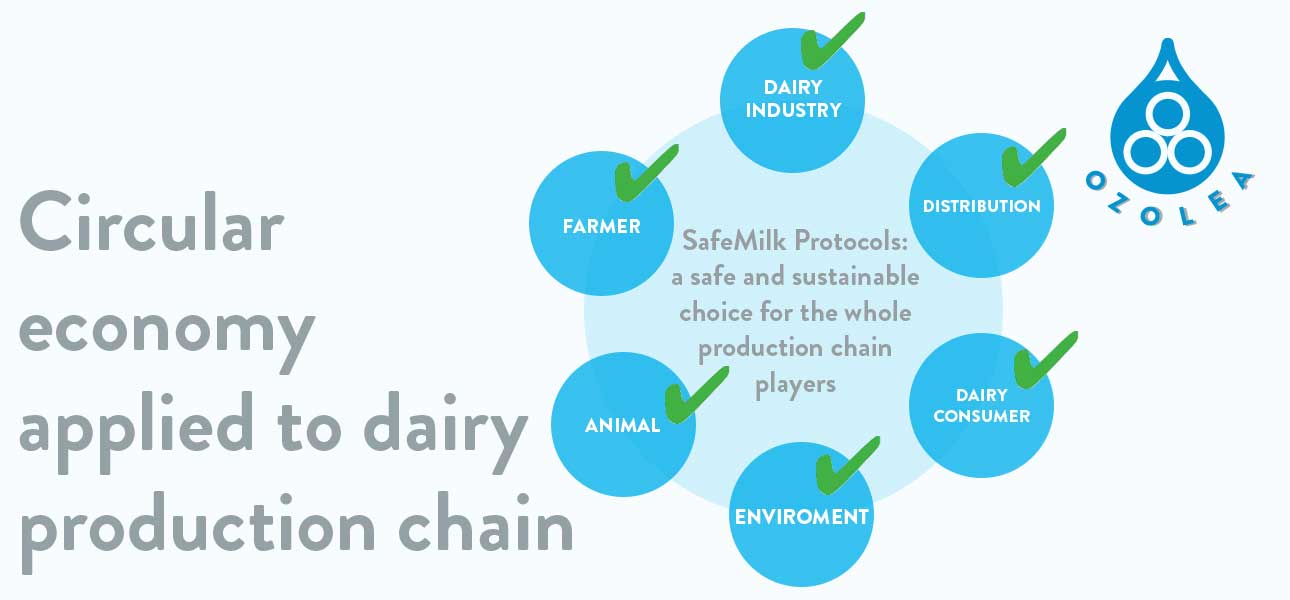
SafeMilk Protocols:
The sustainable evolution of the dairy production chain

For you
Food safety and public health: antibiotic resistance reduction

For animals
Improved welfare and less frequent diseases onset

For the environment
Less food-waste, more drinkable milk with the same resources and same emissions
-
 Why choosing OZOLEA protocols?Instant advantage: at the cost of approximately 2 daysof milk a long-lasting higher production
Why choosing OZOLEA protocols?Instant advantage: at the cost of approximately 2 daysof milk a long-lasting higher production -
 Why choosing OZOLEA protocols?Mid and long term convenience: preserving the tissuefunctionality maintains a high lactation curve
Why choosing OZOLEA protocols?Mid and long term convenience: preserving the tissuefunctionality maintains a high lactation curve -
 Why choosing OZOLEA protocols?Keep control of the delivery/conception rate
Why choosing OZOLEA protocols?Keep control of the delivery/conception rate -
 Why choosing OZOLEA protocols?Reduce the use of non necessary antibioticsand give the best value to your milk
Why choosing OZOLEA protocols?Reduce the use of non necessary antibioticsand give the best value to your milk -
 Why choosing OZOLEA protocols?Our products comply with EU legal framework and are safe for the animal,for the consumer, for the farmer, for the veterinarian,for the pharmacist, for the dairy industry
Why choosing OZOLEA protocols?Our products comply with EU legal framework and are safe for the animal,for the consumer, for the farmer, for the veterinarian,for the pharmacist, for the dairy industry -
 Why choosing OZOLEA protocols?Follow the testimonials of farmers who tried Ozolea
Why choosing OZOLEA protocols?Follow the testimonials of farmers who tried Ozolea

-
 Why choosing OZOLEA protocols?Instant advantage:at the cost of approximately 2 daysof milk a long-lasting higher production
Why choosing OZOLEA protocols?Instant advantage:at the cost of approximately 2 daysof milk a long-lasting higher production -
 Why choosing OZOLEA protocols?Mid and long term convenience:preserving the tissue functionalitymaintains a high lactation curve
Why choosing OZOLEA protocols?Mid and long term convenience:preserving the tissue functionalitymaintains a high lactation curve -
 Why choosing OZOLEA protocols?Keep control of thedelivery/conception rate
Why choosing OZOLEA protocols?Keep control of thedelivery/conception rate -
 Why choosing OZOLEA protocols?Reduce the use of non necessary antibioticsand give the best value to your milk
Why choosing OZOLEA protocols?Reduce the use of non necessary antibioticsand give the best value to your milk -
 Why choosing OZOLEA protocols?Our products comply with EU legalframework and are safe for the animal,for the consumer, for the farmer,for the dairy industryfor the veterinarian, for the pharmacist,
Why choosing OZOLEA protocols?Our products comply with EU legalframework and are safe for the animal,for the consumer, for the farmer,for the dairy industryfor the veterinarian, for the pharmacist, -
 Why choosing OZOLEA protocols?Follow the testimonials of farmerswho tried Ozolea
Why choosing OZOLEA protocols?Follow the testimonials of farmerswho tried Ozolea

World zootechnics on the front line
fighting antibiotic resistance
SafeMilk Conference 2018
October 25, 2018
Cremona International Livestocks Exhibitions
Introduction and moderation by: Mr.Giorgio Setti, Journalist, L’Informatore Zootecnico
ANTIBIOTIC RESISTANCE AND FOOD SAFETY: VALUE YOUR PRODUCT
TOWARDS A DRUG-FREE DAIRY PRODUCTION CHAIN

Luigi Bonizzi, Professor
Universitá degli Studi di Milano DiMeVet
ANTIBIOTIC RESISTANCE MECHANISMS: RESISTOME STUDY IN MILK
Professor Luigi Bonizzi, Professor of Infectious Diseases of Domestic Animals of Universita` degli Studi di Milano and former Head of the Department of Veterinary Medicine and Public Health, investigates the known mechanisms of antibiotic resistance according to the frontier proteomics knowledge and introduces the preliminary study of the RESISTOME in milk.
English subtitles coming soon
Luigi Bonizzi, Professor - Universitá degli Studi di Milano DiMeVet - ANTIBIOTIC RESISTANCE MECHANISMS: RESISTOME STUDY IN MILK
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
I would like to thank Ozolea for giving me the opportunity to clarify an interesting yet complicated topic. Also this morning at a conference there were people operating in the field but with scares scientific knowledge, where infective and contagious diseases were confused and the term infectious was used as a synonym of contagious. These are two totally distinct concepts.
Even when we talk about antibiotic resistance: this is a term that needs to be understood. We are often dazed by incorrect information. Antibiotics have been discovered by a great researcher, a scientist that I like to call a genius: Alexander Fleming in 1928 returns to his lab after a few days of absence and on a slab where the replicated some bacteria he founds a mould, and he notices that around that mould bacteria are not growing. Normally, a researcher would have discarded the slab and replaced with another one. The genius, instead, understood what happened: in that mould there were some active elements that somehow inhibit the grow of bacteria. This has been the first description of antibiotics, that is substances that bacteria and micro-organisms in general use to compete among each other. Micro-organisms have been the first to appear on earth and of course they had to colonize and compete for the territory, environment and nutrition.
These are antibiotics: something produced by bacteria and microorganisms in general in order to create their own ecologic niche and predominate on others.
Nowadays, we call antibiotics some small packages we buy at the pharmacy. In reality, since the very beginning both humans and animals activated defense mechanisms to fight the presence of pathogenic micro-organisms. The immune system is one of these, but not the only one. There is a specific, non specific, and action based immune system that allows the subject, both human and animal, to fight the presence of pathogenic micro-organisms. Today we often hear talking about microbiome: a microbiome is a system of bacteria that colonize a specific area. These bacteria are crucial for the animal welfare: we don’t have to eliminate them all, we need to eliminate those that create problems. Because we do need the others, they are useful as they compete with pathogens producing antimicrobial substances that fight pathogens growth. Therefore you understand these concepts are quite complicated: biology is never black or white, but is made of colors that often blend and create very complex contexts.
Sometimes the immune system is not sufficient by itself to keep this delicate situation under control, and that’s when an animal or a human get sick: at this point, with Fleming’s discovery we have at our disposal medicines that contain this active ingredients, which are active towards other micro-organisms.
Then we can use them in treatment. This is the project: having a medicine that allows me to eliminate a microorganism that creates an alteration, that causes a tissue damage, that provokes a disease. This is the path.
The concept of antimicrobial, instead, is a little wider: we often hear talking about antibiotic and antimicrobial as synonyms. An antimicrobial is a natural substance, that can also be synthetic or semi-synthetic, that kills a bacteria and inhibits its growth. When this can be well tolerated and have a selective toxicity, then it becomes an antibiotic, that can be used in treatment. A disinfectant is a antimicrobial, an antibiotic instead is something that we use in treatment: these are two concepts similar in certain ways but with important differences.
Selective toxicity is the ability an antibiotic has to have a higher or exclusive toxicity towards pathogens, and at the same time not to cause any problem, or a minor and less invasive problem, in the host organism.
Selective toxicity is due to a series of different mechanisms: antibiotics take advantage of the different characteristics given by the fact the bacteria are prokaryotes and the animal or human organisms are eukaryotic. In fact eukaryotic cells dispose of action sites that the antibiotic cannot reach, have antibiotic elimination systems that bacteria don’t have, and display of a series of mechanisms that allow define this type of toxicity as “selective”.
Another important concept is that antibiotics don’t have any power against viruses because in viruses there isn’t any target organ: it must be very clear that antibiotics perform their action within the competition between micro-organisms but they are inactive towards viruses. We’ll see that in viruses there are other mechanisms that we will get to study.
So, when we decide to use an antibiotic we must make sure that we are fighting a bacterial action, because if the infection is viral we are creating an additional problem to the environment, to the animal and to the system. The first thing to do before taking an antibiotic is performing an accurate diagnosis: here the professional, the veterinarian, the doctor play an important role, having to provide an extremely accurate diagnosis. This is the first aspect of antibiotic resistance: in order not to use antibiotics in vain, uselessly, in order not to waste money, and not to uselessly discard antibiotics in the environment, we need to perform an effective diagnosis, based on the principle that if I have a bacterial disease I use an antibiotic, and if I have a viral disease I will use something else.
The introduction of antibiotics in human and veterinarian medicine had a series of positive effects, extremely important: in the farm it guarantees the quality and wholesomeness of products, animal welfare, reduces the spreading of pathogens, has a series of very important positive effects. The improper use and sometimes the excessive use can be for example for a viral infection, where it has no effect, as well when I use it in an excessive dose, or not in the correct way, that is without observing the withdrawal times, or without observing those conditions that researchers recommend with respect to the procedures of correct use of antibiotics.
If I do all this, where does it lead?
It leads to a mechanism of antibiotic resistance.
This because from a genetic point of view bacteria are plastic organisms: being the first organisms appeared on earth, they are able to resist to any condition.
They are extremely efficient organisms, so that if I use an antibiotic they are able to activate antibiotic resistance mechanisms, that is mechanisms that allow bacteria to resist the antimicrobial activity.
The problem of excessive and improper use of the existing antibiotics is of course nowadays an important problem, also because human-animal-environment cannot be seen anymore as independent systems but they do interact: humans, animals and environment share the same micro-organisms in the same environment.
The problem of antibiotic resistance and antimicrobial resistance is an actual problem, by now widespread and perceived worldwide: it’s a time bomb. We need to be very careful and take preventive measures: at an institutional level this is already in process.
What is antibiotic resistance: bacteria are extremely plastic micro-organisms. When I use an antibiotic with the correct dosage, bacteria activate defense mechanisms that make them more resistant to higher antibiotic doses.
Their capacity to reproduce, to survive to the presence of antibiotic is a natural phenomenon that however need to be checked and studied.
How does antibiotic resistance technically occur?
Some micro-organisms developed by themselves mechanisms that block the effectiveness of an antibiotic: they are able to activate efflux mechanisms (they throw it out), don’t let it in, they have antibiotic resistance mechanism of their own. Bacteria are not static, their genome is very plastic and is able to substantially change, to recombine, to acquire genetic material from the outside, this made possible that they acquire certain characteristics and they can transfer them among each other. When a micro-organism is resistant it can transfer the resistance to another micro-organism that is present in the same environment, and it can do this even if it is dead: the genome of the killed bacteria can circulate in the environment or can circulate within other microbic species. In this way they become “acquired resistances” and then “multiple resistances”: a bacteria that directly became resistant to an antibiotic can then become resistant to another antibiotic, for being entered into contact with a genome that was resistant to other bacteria, making the problem bigger and bigger.
However the antibiotic resistance is not a phenomenon that followed the discovery of antibiotics: 30.000 years ago, from a publication of Nature of 2011, permafrost bacteria from 30.00 years ago have been found resistant to penicillin and tetracycline. Therefore you understand that antibiotic resistance is a natural phenomenon: even in our intestine there are bacteria that in order to compete among themselves carry out antibiotic resistance mechanisms that are transmitted through different mechanisms. There are some phages, bacteria and viruses that hit a bacterium and transport its genes to another through complex mechanisms: in our intestine we have an extra logarithm of bacterial cells compared to the cells we are made of (bacterial cells are 10 times the number of cells that our organism is made of). Therefore we carry around a number of pals: in a grain of feces there are more that 100 billions of bacteria and when you take some probiotics you intake 4 billions (kind of useless).
Antibiotic resistance is a mechanism used by bacteria since forever.
Why do we realize now about the antibiotic crisis? Because since their discovery every 2-3 years we discovered a different type of antibiotic, therefore if bacteria became resistant to an antibiotic this was quickly replaced with a new molecule: for a certain number of years antibiotics represented an important tool for doctors, veterinarians and for all healthcare providers. It was an obstacle race where the discovery of new molecules guaranteed to control the phenomenon.
Today, since approximately 15 years, we don’t find new molecules. The problem arose and we started to think about it, considering that these thoughts must keep into account also a productive system based on ethics, more respectful towards the environment, individuals, the productive system, the consumers themselves: therefore it’s a wider and more interesting project and at the same time, helpful from a health point of view. Because we have this problem: since a decade we can’t find new antibiotics, while bacteria are continuing to evolve in their antibiotic resistance.
What normally is not said is that it is true that bacteria activate new antibiotic resistance mechanisms, but if they are not submitted to the selective pressure of some antibiotics, they become sensitive to old antibiotics. We performed a number of researches in the Neaples area with maximalist doctors in urinary tract infections there is an important resistance to fluoroquinolones widely used but these bacteria return to be sensitive to furanes and nitrofurantoin, that is to old antibiotics. To create shared paths of selective use of antibiotics could be a strategy: for the bacterium activating mechanisms of antibiotic resistance is a job that not even bacteria do for nothing, moreover for bacteria that always work in the maximum efficiency, maximum results with the minimum effort. As soon as they lose the necessity to resist to an antibiotic molecule because that molecule has not been used for a while, they also lose the resistance to that molecule and return to be sensitive. In this way it could be useful to create new therapeutic treatments.
In the detail, how does antibiotic resistance occur?
In a population of bacteria, at a certain point we treat with antibiotic and we kill all the bacteria, except for one, that is antibiotic resistant. This resistant replicates (bacteria have a very violent and quick replicative system: every 20 minutes some bacteria can duplicate and that’s how the resistant strain is selected): at a certain point we will have a population entirely resistant. Then we need to increase the antibiotic concentration, but it’s a game we cannot carry on forever. On the other hand we saw that often these bacteria become sensitive again to other antibiotics, and this could represent a strategy: it is understandable that the guideline remains a reduced, targeted and effective use.
Antibiotic resistance is a global issue: in the last 40 years the presence of antibiotics drastically reduces infection caused mortalities, but the risk is that in 2050 we may return to a pre-antibiotic era. This fear spread worldwide lead politics to insist for a reduced and more efficient use of antibiotics, to the extent that in the national chain production starting from January 1, 2019 the use of the electronic prescription will be mandatory. This will permit an accurate control of the use of antibiotics and will allow use to improve and optimize the use of antibiotic. It is certainly an extra effort but it’s meant to create a chain production that is ethic and sustainable: even if it may cause some problem, this is the step we need to take in order to preserve our future generations.
The other important notion is prevention: when we use an antibiotic it’s because we lost, because in some way we did something wrong. Then what we need to do is to bite the time: the soccer player that bites the time usually takes the ball away and this is what we need to do. To work towards prevention, that is to work in a number of situations that allow us to prevent: if the animal is in perfect shape, it’s hard it will get sick, it just doesn’t. If you look at our generation, it has an expectation of life of 80 years, and has a considerable lower number of infections and serious diseases compared to the population living 100 years ago, or to populations exposed to wars, malnutrition, other problems that affect the welfare. It is the same for animals: we need to work keeping in mind to improve the animal management, also through mechanisms of biosafety, improvement of hygienic and sanitary conditions, nourishment, as well as vaccination programs: overall the animal welfare. If you go in an overcrowded room in which you are thigh and bottled up, you feel bad, and when you get out you have a cold and your eyes weep: for the cow it happens the same. The more the animal feels good, the more it succeeds in fighting antibiotic resistance: this is a perspective of less use of antibiotics.
There are then some treatments that are effective and represent an alternative or a prevention of the use of antibiotics: I use them because it’s best not having to have to use antibiotics. Then when the animal gets sick I have to use the antibiotic, and I have to use in a correct and responsible way. Among these systems we find new vaccines, immunostimulants, bacteriophages, antimicrobial peptide, pre and pro symbiotics, new plant derived products, ozonized oils for example, or quorum sensing or biofilm inhibitors.
What is the quorum sensing? Bacteria don’t work each on its own, they talk to each other, they send messages among each other when they need to create a biofilm or harm the tissue and they also send messages to block their replication. It is extremely important to study these mechanisms. Also using bacterial competition can work: we saw that if I have a certain type of lactococcus in the curd, this competes against listeria, which is monocytogenes and in order to replicate and become a problem must be present in higher concentrations in lactoccoccus are present, as they reduce its virulence.
Other mechanisms can be for example the replacement of antimicrobial principles with substances that don’t originate residue, therefore it’s something present in the environment and does not cause any problem. Ozone is a very instable molecule that transforms almost immediately in oxygen, not creating any problem to the environment. Of course it needs to be used in the correct way.
The research we are carrying on in collaboration with Ozolea is to verify the study of the resistome. What is the resistome? First of all the resistome is a study that uses a different research philosophy based on proteomic. Proteomic is the study of proteins that are expressed by a series of genes, that is a genome. The genome is something statical that tells me who they are. It’s like going to the vital record office and get the vital record register: I see the names of all the presents but I don’t get information on what they are doing in this moment. Proteomics reveals if these bacteria are doing something, if they retired or are up to something else.
This morning in a conference it was presented a research by Altroconsumo that examined pieced of chicken breast and found genes of antibiotic resistance, and we are lucky you found them, it’s a totally normal phenomenon on earth: having genes of antibiotic resistance it means that they were there, we cannot know if bacteria with a certain resistance are still there, if they are actively doing something or if they left. We know that some bacteria with antibiotic resistance genes were there, but the research doesn’t tell anything more, and that’s normal. If we consider the bacteria present in our intestine we’ll find tons of antibiotic resistance genes, but I don’t think that we are all currently under antibiotic treatment. Therefore they are two distinct notions: the fact that some genes are present or the fact that there are some proteins linked to the gene. When the gene is expressed, I have the protein. This is proteomics: proteomics searches for the expressed protein. I won’t search only of genes of antibiotic resistance any more, but I investigate if these genes are doing something, if they are performing any action or already retired.
What we saw in this research not yet entirely published for example is that with proteomics we can study those proteins linked to antibiotic resistance genes in a bacterium. I cited just a couple, two paths therefore two proteins liked to two paths of antibiotic resistance. So if we find those proteins that means that that antibiotic resistance bacterium is present. This only from a bacterium. But we are more complicated organism, we don’t have just a bacterium but a ton. In any biological context interactions are complex: there are different bacteria that proceed from humans, animals, environment, therefore it’s an extremely complex environment. And here the term “resistome” is forged.
The resistome is something that takes a picture of those protein linked to resistant genes: the presence of proteins means that those resistance genes are expressed, are working, there is someone who is working for the antibiotic resistance.
What we are doing also with Ozolea is to check if these antibiotic genes can be detectable in the milk and if we can distinguish between resistance genes in farms that use antibiotics and in farms that don’t use them. This is what we did: we checked the antibiotic resistance genes expression in the microbiome, that is among all the bacteria present in the milk. Talking about proteins, therefore expressed genes, not just genes that are present but genes that are actually working, is a totally different thing than checking if the gene is there, that doesn’t mean anything.
I will spare you the method: these are very complex procedures that need powerful database and it takes days to interpret very complex data. What we saw, data not yet published, is that in the milk there is a series of peptides, approximately 8000, 5724 of which belongs to bacteria. I spared you the methodological part: in effects from the milk we were able to select only bacterial cells, from which we evaluated all the proteins that were present, and of these selected proteins, 6000 out of 8100 were specifically belonging to bacteria. 952 of these were linked to antibiotic resistance genes, therefore we found 952 proteins related to antibiotic resistance genes.
I spare you the frequency of proteins involved and the different mechanisms of antibiotic resistance.
With Ozolea we selected two farms in which we applied the system described above: a farms that regularly uses antibiotics, and another that opts for a prevention approach and therefore uses much less antibiotics, if none. What we noticed is that in two similar farms, that use the same antibiotics for therapy, a sample of milk has been collected: in the sample collected from the farm that chose a prevention approach we didn’t find proteins linked to antibiotic resistance. Consider that is a farm: I received this morning data from another farm that uses Ozolea that instead expressed some antibiotic resistance genes, but liked to bacteria or antibiotics not used in the treatment. These are data that we are currently processing, for which some time is needed. Therefore in this case there weren’t antibiotic resistance proteins because probably it was a while that antibiotics weren’t used. In the other control farm instead where antibiotics are regularly used also for dry cows, as you can see we found 13 antibiotic resistance related proteins, therefore there were 13 signals of bacteria which were activating antibiotic resistances: some are due to the environment, that means they are bacteria that activate these genes because they need to compete among each other, but there was also Coli or Pseudomonas which are instead pathogen bacteria, potentially pathogens. So it is clear that the antibiotic resistances we found are all related to beta lactamases, therefore penicillins and cefalosporins. We will go back and recheck to see if in this farm these types of antibiotics are actually used, but it is very likely so. This extremely interesting path allows us to understand and to define a system a little more sophisticated, a little more efficient compared to the genomic study that only gives an idea of the presence of genes.
In conclusion, if we use an approach that allows us to improve the farm and the animal performance, it is clear that we reduce the pathologic conditions that require the use of antibiotics. Obviously through a targeted use and only in pathological conditions and for the correct period of time, we avoid antibiotic resistance and we incur in fewer costs. Everything ends with a sustainable and ethic production.
Chiara Faenza, Manager
Coop Italia Quality Dept -Resp. for Sustainability and Values Innovation
RAISED WITHOUT ANTIBIOTICS COOP BOVINE MEAT PRODUCTION CHAIN
Ms. Chiara Faenza, Manager of Coop Italia Sustainability and Values Innovation Department, explains the evolution of Coop Italia with reference to Coop meats proceeding from animals raised with controlled, reduction and complete elimination of antibiotics, as well as its most recent advancement, starting from news reports that led Coop to place consumer safety, animal welfare and environment at the heart of their production chain choices.
English subtitles coming soon
Chiara Faenza, Manager - Coop Italia Quality Dept - Resp. for Sustainability and Values Innovation - RAISED WITHOUT ANTIBIOTICS COOP BOVINE MEAT PRODUCTION CHAIN
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Thank you for inviting me today, I will take this opportunity to inform you on the production chain approach that we have been applying since 30 years, and that we implemented in past two years on the theme of antibiotic resistance and animal production chains. Professor Bonizzi concluded with an ethical and sustainable production: ethics and sustainability are among our core values, among the cornerstones of Coop’s mission and despite telling how we made this theme a reality, the idea that I would like to convey today is that Coop’s orientation is to set policies and put them into practise, although this is not always a quick and easy task.
I brought back here a title page of an article form the 80’s and an advertising of that time: 30 years ago we had our first production chain products, they were called “made with love”, meat products that evolved over the years, change their appearance, change image, but that allowed us over the time to keep following the Coop production chain of Coop trademark meat products. Actually, over the years we extended the idea of chain production to products not only belonging to the animal production chain, but also vegetal production chain with fruits and vegetables and even industrial products, that we considered and we still do consider particularly sensitive for some aspects.
Therefore an ethical and sustainable production, because yet back then Coop tried to find an healing approach after a scandal, but then preventive for all that followed, because at that time the scandal topic was the treatment with anabolic steroids. At that time the first regulations and scope statements, that did not limit themselves to define the final product characteristics, but that retraced the whole chain production and touched all the possible players involved, in order to intervene in response of our requests as a distributor that doesn’t limit itself to be just a distributor but a distributor with its own trademark products. For us it’s the same as we make those products ourselves, even though we don’t have manufacturing companies, but on which we have a certain knowledge and an important impact.
We started to implement the chain production control in the early 2000’s, these were checks performed since the very early life phases of the animal, primary production both in farm and in agricultural products, to make a comparison; checks on the feeds, feed industries and feed composition requirements, and how these were produced: animal feeding choice and then farming conditions.
Therefore we started with requests that involved the chain production as far as safety measures, quality and environmental sustainability in general. We selected suppliers and farmers: it may seem very simple, it is actually the pivotal point and the most difficult action; we defined strict requirements for suppliers, and since that time forbidden the use of anabolic steroids.
Subsequently, the requirements evolved, always in a perspective of increased hygienic and sanitary protection, and protection towards the consumer, and we defined specific requirements on animal feeding, because taking the concepts explained earlier, if the animal lives better, it is in better conditions, there are better welfare conditions, bio-safety, of course it will give better quality products.
Therefore we requested more control on GMOs in the feeds, we excluded animal proteins, especially after BSE, after weaning, we started requesting welfare requirements, starting with calves and then extending to all the bovines, and we forbid the use of cortisone-based medications. Then a few years ago, we started asking ourselves other questions, stimulated by the frequent press news that were alarming on this theme of antibiotic resistance, because sometimes some problems are left sitting in the scientific world drawers.
When problems hit the headlines it’s when they go under the spotlight and therefore also the consumer is informed on the issue, not always in the correct way. So the World Health Organization, the Italian Ministry of Health, kept alarming about antibiotic resistances and AIFA implemented a number of campaigns and declared its intention to start new ones on the correct use of antibiotics. It has been discussed widely here about the problem of antibiotic resistance, however it was clear that the viable solution was the reduction or at least a conscious use of these molecules.
Just because there were episodes of antibiotic resistance transmission from animals to humans and it was known and ascertained that Italy was one of the podium as far as use of antibiotics in the zootechnical production chain. So we thought about where could we intervene. Not having antibiotics used in human medicine, because one of the main causes is the incorrect use in human medicine, we thought we could intervene on zootechnical chain productions, to control and reduce their use and get to avoid its use when possible, for Coop trademark products. How?
Trying to improve the welfare and bio-safety conditions of all the animals involved. We started from the draft of a scientific report, just because usually our approach doesn’t want to be floating, but to give proof to the media and to the consumers of the seriousness and the need to adopt a correct approach.
Therefore we drafted scientific report that highlighted the problem and set the path for the different production chains. So two years ago we decided to start this difficult path, we launched the project “Let’s raise health”: the project involved different production chains, and as we said in the introduction, the bovine production chain hasn’t been interested yet but it will be soon. We started from the simplest production chains, from the poultry, to the bovines, and then the pork.
The purpose of this project was a change of paradigm: an implementation of good practise, based on these principles of animal welfare and bio-safety, through a responsible use of medicines in zootechnics. We required not to use them or use them only once, just to make them more efficient for the reasons explained earlier.
Avoiding mass treatments, and when necessary, limiting to those cases in which it was strictly necessary, trying to implement a traceability system of the cows, in order to identify those that underwent a pharmacological treatment and those who didn’t, and rigorously only after a diagnosis or a vet prescription. Through this campaign, we reviewed all of our production chains, we selected our best partners, not everybody agreed and we started to request new practises or their implementation with the revision of all the regulations and scope statements.
We redefined the technical scope statements, both as far as requirements and control frequency, both from us and by the various production chains players, while our approach is to raise awareness, involve, that is to work together with the Coop trademark production chain players, and then we obviously redrew our control plans.
This is the timeline that shows the project evolution: we started from the poultry “Fior Fiore”, they were just a few (5) at the end of 2016, to get to all the poultry in Spring 2017; we managed to reach the goal also on eggs and hens; then passed to the pork and bovine, that unlike poultry and eggs that are raised without the use of antibiotics, they actually met this requirement only in the last four months.
This, however, does not liberalise the use of medicines before the last four months: a specific attention, a rational use, are requested also in the previous months. From the meat production chain we moved to industry production chains where these ingredients were used, then all the Coop trademark fresh egg pasta, which proceeds form eggs, hens raised without the use of antibiotics, same for the dry pasta (we already reach the goal but we are clearing out the stocks) and we applied the same logic to products where the ingredient “egg” had a certain relevance in terms of composition, that is in sauces and mayonnaise. The same thought has been applied to cold cuts: therefore with the same logic of the prevailing ingredient today almost 50 types of Coop trademark cold cuts proceed from porks raised without antibiotics in the last four months.
The next steps are other production chains, that might be the turkey ones, dairy cows, or other production chains that for us are more important for production levels. These numbers, regardless the details, want to give you an idea of the size of the impact: it hasn’t been easy because at full speed Coop production chains involve more that 1800 farms and obviously to propose new requirements to the many players required a considerable effort.
On the bovine production chain we required a standard implementation, with reference to the evolution of the scientific progress and sometimes we experimented new ways to work towards prevention. The goal is always to rationalize the use of antibiotics and, when possible, to avoid it. But this is not the main goal as the topic is strictly connected also to the welfare: if the animal needs it, it must be treated properly.
We required and implemented the frequency of controls on residues, both in slautherhouses and in farm. We required the check of medicines registries and we explicitly requested that all the production chains were evaluated according to the CreNBA standard. We implemented the regulation of voluntary labelling, both doing it ourselves and requesting it to our suppliers, because we consider important being able to convey the idea in the simplest and easiest way to the consumer. Later we’ll see some examples of labelling.
Not to create alarmism for the potential residue because this is not the problem, but with reference to the importance of chain production management that precedes the product that they are about to buy, therefore allowing an aware purchase choice.
Digital veterinary certification. We have a part of bovine production that are born in France, and the further the chain production is, the more complex it is to keep it under control.
We implemented a pilot that though a digital system connects the use of antibiotics and or vaccines to the single animal. It is a sort of digital ID easily read and verified, also thanks to an easy to download app and through a bar code reading or RQ code it conveys all the information. The project became real recently, because in September the first animals arrived from France with this sort of digital passport. In this case it has been possible to verify that all the animals have not been treated since their birth. Through this digital system it has been possible to set a vaccine plan that allows to administer the first vaccines in France and the rest in Italy: let’s wait and see if this will be possible with all the animals, if we manage to reach the 100% of animals born in France.
Farm veterinarian and closed-circuit cameras placement.
A couple of yeas ago we demanded a farm veterinarian and for transparency reasons we requested the adoption of closed-circuit cameras in each farm stage: a not easy request that was opposed by many, also for a not entirely negligible privacy issue. When needed we can request to see the videos.
These are some labelling examples: I want to tell you because an intense debate occurred between technical area and marketing on the way to communicate with the consumer because the correct information and education of the consumer makes possible an aware purchase. Therefore we fought because the sentence was “raised without the use of antibiotics” or “raised without the use of antibiotics in the last four months” and not “antibiotic-free”, because in one case it implies an absence of residues while in the other it is highlighted a certain management of the chain production.
I’m done, thank you all.


Chiara Faenza, Manager
Coop Italia Quality Dept -Resp. for Sustainability and Values Innovation
RAISED WITHOUT ANTIBIOTICS COOP BOVINE MEAT PRODUCTION CHAIN
Ms. Chiara Faenza, Manager of Coop Italia Sustainability and Values Innovation Department, explains the evolution of Coop Italia with reference to Coop meats proceeding from animals raised with controlled, reduction and complete elimination of antibiotics, as well as its most recent advancement, starting from news reports that led Coop to place consumer safety, animal welfare and environment at the heart of their production chain choices.
English subtitles coming soon

Renato Colognato, Ph.D.
EU LEGAL FRAMEWORK ON VETERINARY MEDICINES AND OTHER DEVICES: INTRAMAMMARY AND INTRAUTERINE USE
Dr.Renato Colognato explains Italian and European regulations on veterinary medicines and highlights the discriminating factors of veterinary medicines according to the European legislator, as well as the absence of references regarding internal or external use.
UE REGULATION 2019/6 SUPPLEMENTARY NOTE :
The intervention hereby proposed has been recorded before January 7 2019, date of issuance of the EU Regulation 2019/6 entered into force on January 28 2019.
Since the conceptual structure of the new regulation does not modify the core of the normative frame of Ozolea products, the lesson hereby proposed remains valid and therefore made available to the attentive reader.
The Ozolea Regulatory department remains available to veterinarians, pharmacists, farmers and consumers for any doubt and follow-ups.
English subtitles coming soon
Renato Colognato, Ph.D. - EU LEGAL FRAMEWORK ON VETERINARY MEDICINES AND OTHER DEVICES: INTRAMAMMARY AND INTRAUTERINE USE
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Good afternoon all, and thank you for your attendance.
We are going now to focus on Ozolea products.
I would like to thank the speakers who preceded me for explaining what was meant to be a good part of my speech.
My presentation will be more an overview to show that there are indeed products that can be used and are not antibiotics, and of course they respect the veterinary medicine regulations of Italy (ed. European Union).
I am a biologist and I come from human medicine. For years I have been dealing with extensive regulations that of course limit and rule the use of medicines within food chain productions.
Very quickly you will see that there is a strong difference when we consider regulations in the human field rather than the veterinary field, because in the human field we have the “medical device” that is classified in class 1, class 2, etc, and the medicine. In veterinary medicine things are easier.
This does not mean that they are less important, but they are easier because there is a more neat distinction, that is “medicine” or “non medicine”.
It’s obvious, that when we talk about “medicine” or “non medicine” in human medicine there is a series of laws, decrees, regulations that have been implemented, shared with the EU regulations.
As far as veterinary medicine, if you allow me, things are easier because there is only one decree, that is the legislative decree dated April 6 2006 nr. 193 (ed. In EU directive 2004/28/CE), that explains what is considered a medicine in a veterinary context.
As you can see the definition is extensive, I am a biologist not a legislator and I couldn’t get more that a few scientific terms, but the idea of my presentation is to show you how the use of non medicine in a veterinary context, according to the laws currently active in our Country, can absolutely be done without incurring in sanctions. Paragraph 1 subsection a) of article 1, on the definition of veterinary medicine: any substance or combination of substances presented as having properties for treating or preventing disease in animals.
But the fundamental part is paragraph 2: any substance or combination of substances which may be used in or administered to animals with a view either to restoring, correcting or modifying physiological functions by exerting a pharmacological, immunological or metabolic action, or to making a medical diagnosis. Pharmacological, immunological and metabolic are the 3 key words that for us biologist are similar, they have almost the same meaning, because what lays under a direct pharmacological action of a “drug”, meant as active ingredient, is what does the medicine do to the body and what does the body do to the medicine. This is what is considered the idea of pharmacological response, that is then an immunologic response because of course, as Prof. Bonizzi highlighted at the beginning, the immune system is the link between every thing that enters the body and is somehow metabolized.
But in reality, if we talk from a pharmacological point of view, there are some distinctions, therefore we verify that the medicine is actually doing something to the body and viceversa.
This is the idea of ADME-Absorbtion Distribution Metabolism Escretion, that is, the medicine is something that enters, is absorbed, is distributed within the organism, is metabolized, and is execreted.
Hence we verify that these 4 phases are somehow satisfied from a biology point of view, then we check what does the medicine do to the body, that is having an activation target.
This is the idea that in veterinary medicine must be satisfied so that a medicine can be considered so.
This is another interesting slide: the medicine distribution. From a biological point of view that means that suring this process called ADME I can retrieve biological samples and verify that this medicine has been actually absorbed.
In my opinion this is very important: If I have to verify if a medicine had an effect or has been absorbed I have some indicators, called BIOmarkers: the resistome is an example of the ability to verify that something has been modified at a DNA level in the proteic expression and allows me to verify that a control farm actually underwent medicine treatments with antibiotics and another didn’t.
Therefore a medicine can be checked, can be somehow biologically identified and analyzed so that it is absorbed by the body. Hence I can have medicines only for topical use, of which I can find trace in blood.
I can have medicines per systemic use, traces of which I can find in urine because secondary mebolites are excreted in less quantity but we have biomarkers of the medicine and I can follow their path inside an organism.
As Prof. Bonizzi said, this is the classical pharmacological reaction in antibiotics: according to the antibiotic characteristics, this carries out a direct action aimed to inhibit the bacterial proliferation.
Another example that I wanted to make regards medicines with immunologic action. As I said is very difficult for a biologist to distinguish between a medicine that has only a pharmacological effect and not immunological, as the immune system goes along with the hematic stream, so everything that passes by has a direct or indirect action, but in human medicine there are those immunosuppressants that go straight toward certain molecular targets that are in the immune system.
I gave you a very little overview to show that today, in human medicine, biological medicines are specific medicines that have a direct action on the immune system. It is clear that if somehow it can work on a pharmacological, immunological and or metabolic reaction, it’s a medicine.
Of course if we look at the opposite side, that is the non medicine, what do we have? That any substance that does not perform the functions listed in decree 2006 nr. 193 (ed. In EU directive 2004/28/CE), must be considered “non medicine”.
If I have the ability to demonstrate that a substance, an ingredient, a compound is able to indirectly act at a physiological level but without using those parameters: pharmacological, immunological, and or metabolic, in veterinary medicine it is considered “non medicine”.
Let’s take a step back.
Where did we start when we thought that something was effective but non medicine? It’s the classic problem, which is also the vision and the mission of those sitting here today at both sides of the tables: that is, the issue of medicine resistance. I like to talk about medicine resistance because I worked a lot as a tester in the EU Commission, and we worked at the 2011 One Health, and I could list here revisions and revisions that were done, but the interesting thing that I would like to point out is the European concept of “Tackling antibiotic resistance from a food safety perspective in Europe”.
I remember when I used to work in the European Commission, before 2011 they only talked about humans, we needed to improve antibiotic resistance in humans. Then other agencies began to come to the surface like EFSA, EMEA .. the most recent documents that were published are starting to talk about food safety hence the production chain has enlarged.
Today the main focus of One Health it is not the patient as the EU commission thought, but it’s the consumer. The concept of antibiotic resistance has been transferred from being mostly a doctors and biologists prerogative it is now a social concern, an issue that made its entrance in our homes, in our kitchens, and therefore the presence of COOP here today is the evidence.
Who worked with antibiotics knows that in order to work well and use them in the correct way, there are many complications. This is a beautiful review published by the Journal of Dairy Science where they verify the negative aspects of farms that use antibiotics have to face when they choose to work with antibiotics in a safely way, also for the consumers.
I leave you with the table, but I can assure you that the 30-35% of the parameters considered in this review show how they did not get advantages or the animal’s health suffered. In the end who wants to work with antibiotic in an honest and correct way will have to face great issues, if done with a certain ethic.
At the same time another study, likewise published on the Journal of Dairy Science: if I should use the antibiotic in an ethic way I should follow this decisional tree. Fantastic, but I think none of us would like to spend hours trying to understand which would be the best antibiotic, and this makes the whole idea a lot more complicated.
Therefore we found a new concept, both for production and science, that starts from the idea to find a new approach, innovative, I would call it also “natural”, to work in an indirect way and restore what a series of non physiological conditions, both in the environment and intrauterine, that have been somehow unbalanced.
I am very pleased that the professor talked about the microbiome, because of course when we talk about something that from the outside enters an organism we need to deal with what the microbiogen, or all these non pathogen species that colonize our mucous membrane.
Microbiomes are the buccal, pulmonary, gastroenteric and vaginal. When we talk about restoring the microenvironment in non physiological conditions, the mammary is very important for dairy cows but of course we must not forget the intrauterus.
Then the proof of concept, the idea that comes to life with Ozolea for the products meant to restore the mammary and intrauterine microenvironment originates from the idea to find an effective alternative able to work on this balance, that is restore the original physiological conditions of the tissue.
And doing it with topic application on the mammary tissue, working on those can potentially be considered parameters on which there is a pathological condition, there is what we call a dysbiosis, that is an alteration of the microbiome.
The action occurs mainly on the inhibition of the bacterial proliferation for filming effect: this because when the tissues are in dysbiosis, get unbalanced, it’s when permeabilities occur and therefore there is an uncontrolled interaction of all that from the outside (bacteria or other) enters the inside. The purpose of this activity is to re-establish the tissue permeability, through natural regeneration of the epithelial barrier and through filming effect the inhibition of bacterial proliferation.
The data that we obtained to verify that Ozolea-Mast effectively has these characteristics are those that we got on the evaluation of mucous-adesive properties: this is a test performed by third party certified laboratories that demonstrate that these products present this mucous-adhesive property and being made by an oil base they have indeed a filming effect. We are talking about an OECD validated test, therefore of the European pharmacopoeia and it is demonstrated that there is a mucous adhesive ability higher that 17.40% compared both with water and with mucines.
The mucous adhesive property is the first property of any product aimed to the restoration of an equilibrium of the micro environment.
The other fundamental idea is that when we talk about medicine or non medicine, besides the idea of clinical effectiveness, it is the absence of opposed effects or toxicity: its possible toxicity becomes indeed an indicator of its ability to act as antibiotic, hence identifying a direct action.
The products having this mucous adhesive property with filming effect are not cytotoxic: in this UNI EN ISO 10993-5 test we demonstrate that Ozolea is not toxic, that is it does not work as a medicine or as an antibiotic. At any concentration the product did not produce cytotoxic effects. In higher concentrations the cellular vitality is preserved.
Take home messages:
1. Ozolea-Mast and Ozolea-Metr are included in the non medicine cathegory.
2. They are based on food-grade natural components for human consumption, which is a higher level.
3. They do not have side/opposite effects, they do not have cytotoxic effect or direct effect.
4. They do not imply absorbtion residues in the productive chain: prof. Bonizzi will keep showing data but the preliminary are already very interesting, given that the proteomic approach benefits from an outstanding scientific rigorousness.
5. Last but not least, the possibility for these products to be applied both via intra-mammary and intrauterine use: from this point of view the farmer does not risk any responsibility abiding by the present regulations.
Thank you for your attention.
Fausto Cremonesi, Professor
Universitá degli Studi di Milano DiMeVet
USE OF MEDICINE AND ALTERNATIVE PROTOCOLS FOR THE IMPROVEMENT OF FERTILITY AND PRODUCTIVITY. PRELIMINARY STUDY.
Publishing supported by RDP Lombardy Region 2014-2020 - European Agricultural Fund for Rural Development
Fausto Cremonesi, Professor of Obstetrical Clinic and Veterinary Gynecology of Veterinary Medicine of Universita` degli Studi di Milano, describes recent works of regenerative medicine for the improvement of fertility and productivity parameters.
Publishing supported by RDP Lombardy Region 2014-2020 - European Agricultural Fund for Rural Development
English subtitles coming soon


Fausto Cremonesi, Professor
Universitá degli Studi di Milano DiMeVet
USE OF MEDICINE AND ALTERNATIVE PROTOCOLS FOR THE IMPROVEMENT OF FERTILITY AND PRODUCTIVITY. PRELIMINARY STUDY.
Publishing supported by RDP Lombardy Region 2014-2020 - European Agricultural Fund for Rural Development
Fausto Cremonesi, Professor of Obstetrical Clinic and Veterinary Gynecology of Veterinary Medicine of Universita` degli Studi di Milano, describes recent works of regenerative medicine for the improvement of fertility and productivity parameters.
Publishing supported by RDP Lombardy Region 2014-2020 - European Agricultural Fund for Rural Development
English subtitles coming soon

Chiara Turelli, Engineer
OZOLEA President and CEO
PRESENTATION SafeMilk PROJECT
Ozolea President declares the scientific mission of Ozolea towards a reduction of antibiotic resistance spreading in food production chains and, through the SafeMilk project, how this goal can be an opportunity for the whole milk production chain to build a safe, healthy and sustainable future.
English subtitles coming soon
Chiara Turelli, Engineer - OZOLEA President and CEO - PRESENTATION SafeMilk PROJECT
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Thanks to the journalist who accepted to be the moderator in this meeting and thanks to all of you, your presence is very important.
Ozolea is a scientific reality. We have been working at this project for almost 15 years: we chose to show up on the market with a commercial product three years ago, at the beginning of 2016, and we did it because some farmers who experimented our product with us and with professor Cremonesi during Dr. Andrea Bignotti’s doctorate thesis, were insistently asking for the components of our products. They used to say “at least the simplest” and the simplest was this “bioengineered” oil, as we use to call it, because we modify the oil structure and we made possible that this oil gains particular characteristics, among which the absence of pharmacological and metabolic activity, for the reasons already explained.
In this context we decided to take on the market to give the farmers an harmless tool. But we’ll get here. On this occasion I would like to present you the project SSafeMILK that we are launching these days at our exhibition stand and awarding 50 paragon farms that through these three years reduced the use of antibiotics: you can tell from the veterinary medicine registries, the number of animals on farm has nor decreased but the overall quantity of intra-mammary antibiotics purchased and registered did.
The product itself was not sufficient: it took a great ability on the farmer’s part to know how to use it, how to apply it. This is not a commercial experience tied to just one product, but it’s an experience of a farming model. Without this ability of the farmer it is not possible to reach a null or closed to zero antibiotic resistance result as defined according to the resistome project. The farm presented earlier is a farm that has been using Ozolea-Mast both in lactation and dry for the past 3 years.
For this farm it hasn’t been easy to start this path: there were many doubts and many linked to the costs because the approach mentality to the farm that needs to change, and it is necessary to reconsider the numbers upon the results, which are not just the results within the farm but results considered within all the production chain and therefore promoted.
And it’s for this that we launched the project SSafeMILK.
The project SSafeMILK is most of all a food safety project. It’s a food safety project not according to the parameters set by the law today, because this is law and it cannot be the reference for the future. The reference for the future is the scientific research, the scientific avantgard puts us in the conditions to have new instruments. Are all of them reliable? We will find out together.
We cannot say we have an univocal scientific solution in our pocket, that puts us in the condition not to listen to all the rest.
It’s an economic sustainability project: the farmer must be helped to do the math in a correct way, to find those margins, and we are convinced there still are many, of savings, waste reduction, that create distortions throughout the whole production chain.
It’s an environmental sustainability project, because, making it easier, when we say “we reduce the waste” it means we are going to improve the resources use parameters in comparison with the milk produced and saleable.
It’s a project of fight against the antibiotic resistance, not in a way that makes a flag of it for marketing reasons: it is needed an honest information on that antibiotic resistance so deeply modified in the latest years by an aggressive chemistry, leaving alone the natural antibiotic resistance that is part of a biological process.
Therefore it is a project of social responsibility mostly. It is a project of social responsibility on the whole production chain.
I was smiling earlier when Ms. Faenza was talking about the article from the 80’s. I was very young but I do remember some topics discussed at home: my father sue and won against a feeding industry giant because there were anabolic steroids in their feeds for the calves growth. We won the lawsuit and the reasons were correctly presented also by the defendants, but however my father was not selling to the final consumer, and the fact that this wasn’t our core business but a side sale compared to the rest of the business, this sense of having betrayed the consumer that he did not see in the face because he was selling to a merchant, and there was a production chain before getting to the person that would have eaten that steak, and I remember this sense of having betrayed the consumer as a failure. This sense of failure belongs to the production chain, not to the single individual, it’s also who sold that feed, who formulated it, it belongs to everyone and leads to negative results that sooner or later will harm everyone, also the satellite activities, also who is behind, also who makes the bolt of the feed mixer.
I insist on this idea because it is the whole production chain that profits form the milk production: maybe it’s the case not to point the finger or saying it’s someone’s fault, maybe it’s best to work together towards a solution.
This is what this project SSafeMILK wants to be. Today it isn’t a predefined project: we are building it together, also based on the needs of all the players involved in the production chain.
Someone in this room and many outside know that Ozolea during these years truly phone called the world: we called everybody, we built a network, someone was bothered because it wasn’t clear that Ozolea comes form a scientific reality and we looked like those who sell the latest product, it didn’t looked like something so well structured.
But we gathered a considerable amount of information, we collected the needs of this production chain, we collected the worries of veterinarians, who are requested to adopt protocols of antibiotic reduction in farms and must find alternatives: they cannot be left alone looking for alternatives, that cannot be the usual “fresh water”, but it must be something effective and conveyable to the farmer, because the collaboration with the farmer is essential.
We tried to involve pharmaceutical industries: many did not listen to our invite, other listened but nothing more than that, but maybe there are still some possibilities to work together. These have a fundamental role: it was reminded this morning at the conference AISA that the pharmaceutical industry needs to have a market guaranteed so that to have margins that permit the research; we depend on a research that must continue and with the right funds. Maybe all the participants somehow owe their life to a medicine or an antibiotic, that if didn’t exist we would be mush less in this room. Therefore we kept into consideration the pharmaceutical industries even though someone dare to tell us that we break the market: we don’t break the market, on the contrary, we want pharmaceutical industries to maintain their financial volumes with less volumes in term of kg of medication spread out.
We involved the pharmacists, because on the market we find many products liberally sold that it is not clear how they are classified. And for this reason we did our researches, and what is available is decree 193/2006 (ed. directive 2004/28/CE), we read it word by word, we studied it, we verified with reliable sources, not local interpretations as unfortunately other people did and who influenced them.
Obviously we involved the farmers, or better we are going to award them in these days because the award we want to give them is a social responsibility achievement, because they understood the model, they understood where they benefit using a product like Ozolea-Mast for the antibiotic use reduction. Someone wanted to eliminate completely the use of antibiotics, but if it was really necessary they would buy it instantly on the vet’s recommendation: we don’t intervene in the disease concept, but we try to make possible that the animal maintains a welfare condition, that does not get a disease, as far as possible.
We tried to involve the dairy industry, and shortly we’ll see why.
And last, we care about the consumers needs: we are consumers ourselves, we are people with the society macro-needs, including environmental needs, that for me are also a sort of professional deformation and it is a theme particularly dear to me.
We contacted then everybody, and collecting the various needs we arrived on the market with these two solutions, two apparently simple products, but which are not the cornerstone of the project: the cornerstone of the project is the protocol, and the farmer’s activity who needs to learn how to better manage the welfare status of his animals. We do it giving it a name: we called it SSafeMILK and now I am going to explain you what is the project SSafeMILK, that is what we are going to do because we consider this a starting point and let’s see together what we want to implement in the next few months in order to have a better expendable result in a timeframe of 12 months maximum, hypothetically.
SSafeMILK project is based on a demonstrative project that consists in 3 panels:
– Health and welfare, where we talk about parameters that go beyond what is defined today as health and welfare, therefore we will keep into consideration the preliminary study presented by professor Bonizzi, and we will conduct a study on large scale. We needed a preliminary study to earn interest, and we’ll see if this will be the case, public funds to validate this system and collect valuable and expendable results. We will conduct a study linked to pharmadynamics, more for investigative desire because as we’ve seen we don’t have a pharmaceutical action, but we do it to grant the institutions, and moreover EU institutions, numbers with which doing the correct and relevant evaluations, most of all to push for the issuance of this regulation on the veterinary medical device, in which our products today would be placed if there was the law.
We do it to collect clinic parameters to define the best animal welfare: this morning at the meeting I was referring to they were saying that the perceived quality of an aliment is first of all its food safety, secondly where it comes from, and how healthy are the animals that produced it, and third it’s actual goodness. There, we want to collect welfare parameters that go beyond what has been done until now and clearly we open our collaboration to those who have good ideas and we widen our work groups just for our scientific nature.
– We will also evaluate the food safety and suitability: here we want to involve the dairy transformation industry, not only for parameters linked to milk for human consumption but also for parameters linked to fresh cheeses, average and aged. We will perform analysis in collaboration with the transformation industry because the transformation industry received lately milk that sometimes did not have those characteristics necessary for the transformation that should instead be guaranteed, to discover later that the farmer used new medicines, or new products that, although absolutely legal, apparently shouldn’t have left any trace but actually inhibit certain types of transformation.
Therefore, to avoid getting to this point, having wasted so much time, so much money, such great effort, according to a purely financial calculation but that clearly has its effects also on the environment, and has a social cost, we propose to analyse this first, to build together the protocol, to evaluate together these parameters to stop those solutions that may not work for somebody. We want a solution that could work for all the production chain players. In this terms I think as an engineer and I say: let’s systematize this, it will be more difficult and require more collaboration to get to a solution, but if we get there together we will obtain a solution that will allow to resolve more cases and stay more efficiently on the market.
– And we want to do an environmental evaluation: we do it both on the product Ozolea, where we have only one ingredient which is vegetable oil suitable for human consumption, although with particular characteristic. We do it because we want to reduce the environmental impact of the product itself: this is just the beginning and there will be an evolution, which is almost ready but we are in a path that allows us to be particularly suitable for the needs of today’s society. But we want to perform and environmental impact evaluation according to these new parameters also of the farming model: we will check how much CO2 has been saved by the farmers who used a certain number of Ozolea-Mast or Ozolea-Metr applications instead of other products, how the resources were managed, after all an animal has its own environmental impact and if I optimize its production I obtain a particularly positive balance.
And then what do we do with all this data, all this information that we built? We build a standard, a certifying model that is directly expendable to the consumer.
I am going to present you a small sample of out sustainability chart: it’s the sustainability chart of Ozolea-Mast but it can be perfectly applied also to other protocols.
The farmer convenience is an immediate convenience, because the farmer does not discard the milk in the days when the product is administered, therefore I optimize at least its financial balance. Then there is a convenience in lactation: an antibiotic has the respectable effect to kill the pathogens and it’s an emergency solution: if I want to support the tissue functionality I need to intervene before the emergency.
If I kill the pathogen and I don’t do anything to support the tissue functionality in that difficult quarter I will have a decrease in the lactation curve. All the farmers say this: if a mastitis occurs on a quarter, you can save the quarter affected but the part gets wasting and it will never produce as the other three. And still that animal will eat as an animal with four quarters, and will produce greenhouse gases as an animal with 4/4. Then let’s consider: if I can support the functionality of the apparatus, even if indirectly, giving the best ingredients to the animal so that it can autonomously defend itself, then the farmer will have a much more valuable advantage, not only on this one but also on future lactations.
Ozolea-Mast has this advantage of antibiotic resistance reduction, that antibiotic resistance induced by the excessive use of medicines, because luckily antibiotic resistance is reduced with the simple reduction of antibiotic use. And this is a mechanism of which we need to take advantage now, because we don’t know how these mechanisms will evolve in the future if today we ignore them.
We are talking of less stress for the animals because we maintain a welfare situation before the outburst of a disease.
And we talk about better parameters for the evaluation of the animal welfare, as we already said.
We talked about less resources used and less greenhouse gases produced for saleable liter of milk. And finally we talk about a lower environmental antibiotic resistance: animals are in contact with the environment and when you use antibiotics on them, we were talking about the idea health-human-animal, also this morning the idea has been widely explained and I feel I want to add that sometimes we forget we are mammals and we belong to the animal kingdom.
Therefore we collected different players and player groups of the production chain to which some sections of the production chains are significantly linked to, like
– animal health and public health linked to veterinarians, pharmacists, pharmaceutical industries, or
– the idea of shared benefit between farmers and veterinarians: the idea of farm veterinarian promoted by the new program of the Ministry of Health is essential and I believe the veterinarian should be widely acknowledge in this role of protector of the public health.
– The idea of food safety, of which the dairy industry is a grantor at the eyes of the consumer.
Today we invite all these entities to show their interest: many already did it, we are collecting subscriptions to this project.
It will be a project in which who is there first, will enjoy the results first; who arrives later will simply find something done to offer to its market. For sure nobody will ignore this type of approach, be it the SSafeMILK project or another project of improvement of what is proposed to the market today.
This is the SSafeMILk project that I wanted to present to you and which I hope you will agree on.
We conclude with our usual wish “have a good milk and savory cheeses”!
I invite you to stay in touch and thank you for staying until the end.

Interviews and awards SafeMilk 2018
Cremona International Livestocks Exhibitions
English subtitles coming soon
VANZETTI HOLSTEIN - How to reduce by 60% the use of antibiotics in lactating animals
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Q. Welcome back to the SSafeMILK interview round towards a farm with no medicines. Directly from the 73rd Cremona International Livestock Exhibition today we will present the experience of a major dairy farm, a reference point, mostly among farmers with a passion for genetics, who attend Holstein breed shows. We have here with us Mr.Marco Vanzetti, from Vanzetti Holstein farm in Tourin. Good morning Marco.
A. Good morning everybody.
Q. Thank you for accepting our invite and for being available to present your farming model. You have an exemplar farm, that has been always taken as an example by many other farmers, is that correct?
A. Thank you for the compliment, we try to do our best and also we try to share as much as possible what we do, both our mistakes but most of all those things well done, that can be useful to everyone.
Q. It is certainly a very open approach, useful for a production chain constantly trying to improve. Right?
A. For sure, the dairy production world in Italy should work in this direction, trying to work together towards a concept of quality, an idea of promotion of an excellent product as the Italian milk is.
Q. We are glad to hear this, and this is exactly what we are trying to do, we started in a very simple way from scientific research and we are presenting it in these very days of exhibition with the project SSafeMILK. We will have a chance to examine it in deep and we’ll get to the end of this meeting with an award ceremony that acknowledges to the bravest farmer the merit of being open minded. But for now, I would like to know something about your farm: tell me how it is organised and how you make your choices in such an articulated field.
A. Our farm is located in Candiolo in the outskirts of Turin, about 7 km from the Piedmotese main city. It’s a family business, with myself, my brother Davide, my sister Serena, and with the help of our parents and two employees we manage the Vanzetti Holstein. Since 2016 in our farm we have been working in AMS, with robotic milking, in a new facility of 250 milking cows capacity, along with fattening animals for meat, and two biogas plants for the production of 2 megawatt. Our choice to switch to a full robotic milking in 2016 actually started a few years before; in fact already in 2010 we had a group of animals in robotic milking, and we passed to this milking type for all the animals in 2016. This milking type has many pros and a few cons: the management of mastitis was one of the most complicated aspects to keep under control, and this has been one of the reasons why we decided to try Ozolea technology.
Q. OK then would you like to tell us for example which have been your doubts, especially at first, choosing a product like this; if you had any difficulty to get used to the protocol, building your own protocol. What can you tell us?
A. Also in the past we tested other products based on the same theory, on ozone antibacterial properties, but there always were problems with the product preservation, with the product syringeability, and also problems with their effectiveness in general. Nevertheless we kept believing that sooner or later we would have been able to employ the ozone properties to reduce the use of antibiotics in farms, and having good results as far as milk health. Moreover, in a system like the one of robotic milking in which the habit of milk separation generates actual losses in performance of the milking structure, the use of a product like Ozolea allows to get there in advance, that means to decide to intervene also when there aren’t such clear symptoms and not having risks or losses for discarding milk, and you can also have a support for preventive therapies, or if like us you use bacterial slabs protocols to identify the bacterial agent, with Ozolea you can start and work immediately, don’t discard your milk, and then you can eventually pass to an antibiotic therapy, even though what you are already doing turns out to be more than enough.
Q. That’s great, so you basically built your own protocol also starting from the technology you adopted. Let’s consider the fact that Ozolea-Mast doesn’t have a specific pharmacological effect, it doesn’t have any pharmacological effect at all.
We carried on a research that allowed us to obtain a non medicine product, therefore that does not have any pharmacological or metabolic effect, it does not interfere with the animal physiology, but it does a specific job in this very moment: when you see the first signs of unbalance in the tissue we support the tissue functionality and productivity.
And being able to realize it in this moment, dealing with somebody who can do the math properly in his own business and who acquired a technology that is able to detect as much data as possible and is able to interpret them correctly, support the tissue functionality and therefore the lactation curve, allow us to achieve a relevant saving and most of all to avoid a huge waste. Can you give me a general idea of the numbers that brought you to embrace this type of protocol?
A. In the past 24 months we reduced the use of regular antibiotic tubes for mastitis by at least 60%, and this both by switching to Ozolea tubes and working on the identification of the bacterial agent. Knowing that when you start doing a job with ozone chemical-physical properties you are nevertheless doing the right thing on the quarter that is infected in that moment. Then, if in the 24 hours in which you have the result on the bacterial agent you are already having a positive feedback then it is convenient to continue with Ozolea. If instead the symptoms tend to worsen, then the use of an antibiotic might be necessary, but the quantity of antibiotics use gets reduced, you reduce the resistance risk and increase the effectiveness of those few antibiotic treatments.
Q. Therefore you are telling me that you use it in lactation. Did you had a chance to use it also for cows in dry or not yet?
A. For now we are using it on cows in lactation also because for dry cows we are trying to reduce the treatments to the minimum, we only work with the sealer. But why not? We could also try to use Ozolea. In my opinion, what I would like to do with those animals who have some somatic cell too many is to identify the quarter infected and treat that one with antibiotic while the others with Ozolea, to help them with Ozolea bactericidal action.
Q. Ok, Ozolea does not have a direct bactericidal action: I want to remind this because here lays the difference between medicine and non medicine. When we studied this type of product we truly intended to provide a tool with no side effects to the farmer, something risk free that could be used safely. Let’s consider that it is not necessary to use it in dry of course, everyone adopt his own protocol and actually your global attitude for a reduction and conscious use of the medicine and of the antibiotic in general is a winning one.
What we would like to do with this SSafeMILK project is to have also other production chain players could realize that there is a change underway, a change of awareness, and to have them willing to spend a little bit more for a product that is generated from criteria derived from what today the law rigorously requires. What do you think?
A. For sure when we talk about valuing the Italian product, it is not enough that the farmer work better, but also the work done must be capitalized someway. For sure when you work well you increase also the business income. But for sure this increased quality and increased effort must be acknowledged.
Q. Very good so with the presentation of this SSafeMILK project we started to create interest. Yesterday we invited the transformation industry, that a big player of the large-scale retailing sector accepted our invite to consider the feasibility of this project.
We extended the participation at this round table for a redefinition of a milk price, based on the new voluntary certification parameters based on the SSafeMILK project, today we are satisfied with an award mostly for the social responsibility model towards antibiotic resistance, for the animal health and welfare model, for the financial sustainability of zootechnical productions that cannot be less, but also for the environmental sustainability: if we reduce the waste we reduce the impact of the zootechnical business on the environment, and this must be conveyed to the consumer, it must be acknowledged and it is necessary to relieve this weight that too often the dairy industry and the whole milk production chain get on their shoulders, also in terms of confidence in the production chain. Therefore we award the Vanzetti Holstein farm for this commitment and most of all for this innovative approach. We wish you the best, obviously this award will be followed by a series of products: we will offer the well-known Ozolea-Mast, as well as our new product Ozolea-Metr. We’ll be waiting to hear from you about this new product and we thank you, looking forward another appointment with something more expendable for the industry.
A. I would like to thank you for this award, for the possibility to be interviewed here but most of all thank you for your desire to find a different path to solve everyday problems. I am sure this clean route, this green route can bring advantages to all the farms but also to all the satellite activities.
Q. Yes, absolutely, let’s say it is a mutual thanks because “it takes two to tango”, and when you share big goals you risk to get there!
A. I hope!
Q. I hope!
A. Thank you!
GUIDO OITANA - Welfare or treatment? The more productive choice
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Q. Good morning, welcome. Directly from the 73rd Cremona International Livestock Exhibition, we are starting the SsafeMILK interviews cycle, towards a farm with no medicines. We will present here some experiences of farmers who are using Ozolea products to reduce the use of unnecessary antibiotics on farm and we start from a farm that represent a point of reference in the dairy farm business. We welcome Mr.Guido Oitana who breeds in Scaleghe, in the province of Turin, and often takes part in national and international exhibitions. Thank you for accepting our invite.
A. Thanks to you.
Q. We know you are in show with your animals also here in this exhibition: can you tell us what did you bring?
A. I brought four cows and three heifers: some of them have already been shown in other exhibitions, two primipara and two first time.
Q. For sure we expect to win something because usually your name gets to the podium. And we would like to tell a little what was your experience with our product, but first we would like to know how is your farm structured. What can you tell us about this?
A. We milk 200 cows free range with free individual stalls, and we milk three times a day. We started with three milkings three years ago, we are very happy with that and right after having started the three milkings I started with the selective dry, that is to treat only the cows with higher somatic cells with antibiotic in dry and the others were treated only with cap to protect them during the dry time.
Certainly, one of the most positive aspects of the third milking is the mastitis onsets reduction, which have been almost halved.
Q. We would like to give a more technical angle to this meeting, because we know that many are complaining that the zootechnical sector is a tangle of different expertise: they say the farmer must be an expert in a little bit of everything. In your farm, how are important decisions taken? Such as the third milking or the animals management?
A. I count also on professionals, like our veterinarian, who is a very good one and it has been years now that with the veterinarian we try to prevent diseases, not to treat them. The veterinarian on farm should not profit from the farmer’s misfortunes: in fact, joking, he always tells me “ you should pay me for the twisted stomachs you don’t have, rather than for those you have”. Practically we pay for a sort of subscription to have his consultations, therefore, intervention on actual diseases are rare, because this is one of the wrong ideas at least in my area where the veterinarian is seen as the one who treats sick animals. On the contrary the veterinarian must be a farm consultant that makes sure the animal does not get sick. My first goals when I started to be a farmer were to have the maximum production, then when you have the maximum production problems increase, also because you are not good enough therefore the second goal I set was the animals health, which is one of the most profitable things. With the production I reached good results, even if with more problems, then we decided to aim to the maximum health, and this is almost more profitable than the production itself.
Q. Then we are getting to the topic that we set for this interview, that is welfare VS treatment: the more profitable choice. You are telling us that keeping your animals in a general welfare condition allows you to avoid problems and to have higher productivity.
With this respect, would you like to tell us something on your decision to use Ozolea-Mast?
A. Yes, I got to know this brand I think three years ago, you had a stand in the other pavilion then I thought I could do something because both through reading and from my experience with traditional antibiotic treatments, [I know] you may resolve a percentage of mastitis around 50%, while if you leave things as they are they resolve by themselves in approx the 30% of cases, then your treatment increases the results by 20%, and it doesn’t pay back to have the 20% more of recovered animals. Therefore I decided to try a product that would allow not to discard the milk, and now I am also more convinced as in our area some dairy factories started to perform different tests for antibiotics on milk, there are some tests that are more accurate than the one prescribed by law that still find antibiotics for which it wasn’t necessary to withdraw milk. And not only antibiotics but also anti-inflammatories at zero withdrawal are found in the milk, especially when the milk is transformed into cheese it creates long preservation and it creates problems later.
This topic is critical with the industry, as us farmer we are right, because having been declared the “zero withdrawal” we followed the official instructions and we are on the right side, but the industry still demands the withdrawal for products for which it shouldn’t be necessary.
Q. Let’s say it’s a reality we know very well, because within the production chain very often many responsibilities are put on the farmer’s shoulders, even though technically the farmer simply respected the rules that were imposed. In this situation you presented some very important themes: the food safety, the fact that the product must be practically perfect in terms of safety for the consumer, the theme of public health linked to the antibiotic resistance mechanism, of many antibiotics that are used too often, besides the protocols of true necessity.
In this sense we say that to keep the animals in a welfare condition allows to have more productivity and therefore more profit for the farmer. The dairy farm, the dairy store they tell me adopted the protocol to use these accurate tests, do you think they are going to recognize a value, a surplus value for that milk that is produced with more rigorous protocols without antibiotics or are they still far from this?
A. Well, I believe that the relationship between producers and industry has always been difficult: it’s not a production chain relationship. I believe they will work to change the official laws. They are certainly moving in that direction and however besides that it’s up to us to fit, change a little the way we work, because it is typical of us farmers to find shortcuts or use anything, so that the milk does not come out positive to tests.
If there were some statistics, some products are certainly over used and the farmer pays for them, but only who produces them profits. The use of medicines in farms has been already reduced a lot, but I believe there is still a long way to go. We are working with a group of farmers with unions to create a regulation on the responsible use of antibiotics in dairy farms.
It’s a complex topic, it’s a cultural issue and its solution is not easy or not easy to apply, even if we will be required to do so. We think we should do it voluntarily, but we know that sooner or later the dairy industry will impose it to us.
Q. As for all the difficult issues we start from the awareness of the issue, we gather around a table to find solutions. This is what Ozolea did throughout these months launching the SSafeMILK project, that intends to acknowledge also financially the farmer the advantage for this effort, of making a choice that goes well beyond the parameters set by law for food safety.
In fact, in this interviews cycle we will be awarding, we are awarding some farmers, a lot of farmers, approximately 50, most of all for their social responsibility in the fight against antibiotic resistance. Therefore I pull out your award: Oitana Guido and Ezio Farm from Scalenghe has been using us since 2016, we award Mr.Guido Oitana for the social responsibility model towards an antibiotic resistance reduction, for the innovative approach as far as animal health and welfare, for the financial sustainability of zootechnical productions because adopting a solution like Ozolea-Mast we considerably reduce food waste, therefore for environmental sustainability because you considerably reduce the use of resources and greenhouse gas per liter of milk produced.
Therefore I confer you this award, you know this award will be followed by a series of products that will be sent you home. In the meanwhile, Mr.Oitana, I let you go and check on your animals and I wish you to win also this time.
A. Let’s hope, let’s hope we’ll be lucky we have a Piedmontese judge, we don’t know if it’s going to be an advantage or disadvantage.
Q. We’ll find out on Saturday then, thank you so much and see you soon!
A. Thank you.
ALL.GANDOLFI - Ozolea-Mast suitable for Organic farming
©This work is subject to copyright and it is an exclusive property of the Author.
Please be aware that disclosure, copying, distribution or use of this document and the information contained herein is strictly prohibited unless with written authorization by the Author.
Contact Ozolea to get in touch with the Author.
Q. We’re back to the SSafeMILK interviews cycle, towards a farm with no medicines. Directly from the 73rd Cremona International Livestock Exhibition, we now finally introduce you a veterinarian, Doctor Gandolfi, who owns a farm and delivers milk for organic Grana Padano in Piacenza. Hello, we ask you to tell us about your farm and how it is organized.
A. We have a dairy farm, we deliver our milk to a social dairy factory, we recently became “organic” and it has been for this reason that we discovered your product, and we opened up to a new horizon and a new adventure. We have 350 cows, 300 of which in lactation and 50 in dry, plus all the heifers. We had a room with a carousel, we have now been starting with the construction of a robotic milking system, so we are starting also this new adventure. Since we discovered your product we also started a project with the zooprophylactic institute of Piacenza for the control and a targeted approach for mastitis and therefore also the use of antibiotics on farm. Most of all, as far as concerns clinical mastitis, we are taking part in this project and we have the opportunity to slate testing (CMT) the milk, therefore consider the targeted use of antibiotics or not. Your product has been very useful for those uncertain moments, or for subclinical mastitis, or for the very early stages of mastitis in which we mainly use your product.
Q. To this extent, we remind that Ozolea-Mast is a non medicine veterinary product, therefore we don’t have to expect that pharmacological effect or that metabolic absorption that characterizes all those products that are in fact a medicine, and maybe also particularly effective, for example one-shot products. In fact this is a product that supports the tissue functionality, and it is necessary to be patient in getting used to it. Was it hard for you?
A. At first yes, maybe because we did not understand how to use it and our expectations did not match with the results. But that was because they did not explain us which were the steps: we were expecting a sort of homeopathic or phyto-therapic medicine, that immediately reduced the problem. We also did a mistake on the action speed: your product works really well if used in those cases in which mastitis, or the problem, is caught at the right time. If the problem is dragged around, obviously the effect is not as strong.
Q. Sure, we tried to emphasize the importance of the prompt use, encouraging the farmer not to wait the symptoms of a full-blown mastitis but to intervene on first signs of tissue unbalance. For this very reason it is important to remind that since it does not have a pharmacological effect we cannot expect a miracle product, or a miracle solution from something that does not have an active ingredient.
Basically Ozolea-Mast builds a barrier that protects the tissue and allows the tissue to have a break form the bacterial attack, a temporary relief, and facilitates the regeneration of the tissue itself. In this way, being the very same animal regenerating and rebuilding the possibility to defend itself autonomously, it is necessary to be patient and completing the eight applications cycle. Did you find other problems, other negative aspects, I don’t know can you tell me something about the costs for example? What did you think?
A. OK we have to consider that for the type of product the milk can still be sold, and on the other hand the cost of a mastitis if caught later. We need to start form an assumption: if we have a certain organization, that if I detect the problem at a later moment I still have in my hand something that helps resolving, then the medicine quality and efficiency gets lost. We need to have a good timing, good product in our hand and start from there, evaluate the mastitis to resolve the problem. If instead we keep thinking we can treat something that slipped out of hand, then it’s not OK. Your rule is correct, that is to intervene promptly on the problem: resolving the problem quickly of course I waste less milk, I waste less product, the bovine is not affected too much, and therefore we have the profit. And the animal health pays everything back.
Q. Yes, in fact to keep the animal healthy before having to manage health issues allows us to keep a high business productivity not only from a financial point of view, but also in terms of animal performance, and also, allow me, in environmental terms: we are going to have a reduced environmental impact and a productivity able to satisfy the food consumer requests, with a lower use of resources. Do you know that we are setting a SSafeMILK protocol collecting the experience of farmers who used Ozolea-Mast in these years, and are starting now to use Ozolea-Metr, our last product for intrauterine use: we are doing this because we are sure that within the production chain there are still huge externalities that need to be codified and which value must be redistributed within the production chain to the players that put that extra effort to obtain some results. Usually on the farmer’s shoulders there is the responsibility of many choices; we were saying the farmer must be an expert in everything. But you have the virtue of being a veterinarian: how are decisions taken in your farm? How did you organize the decision team on big changes, also the decision to switch to organic?
A. The decision to switch to organic was maybe originated first of all on the farming, I mean crops and the fields management. We also have biogas and we saw that we could work well also outside of traditional methods. And from there we started our path, first in the fields, then little by little we saw that an organic animal farm was feasible. We will keep the other traditional farm as is, because we also want to see the difference in the management; also if you throw yourself in an experience it becomes hard to evaluate the differences.
Q. Yes, absolutely, it’s a very methodical approach.
A. Therefore we are keeping the two farms, we are happy with both and we keep going, we never stop.
Q. And we will meet again next year, reviewing the results.
A. And I’m curious to try Ozolea-Metr.
Q. You will find Ozolea-Metr in the prize package that we prepared for you and exactly for the social responsibility benefits in the fight against antibiotic resistance, food safety, animal health and welfare, financial sustainability in the zootechnical sector with the adoption of the Ozolea-Mast protocol, today Ozolea wants to reward you with a certificate and a set of products: you will receive Ozolea-Mast and Ozolea-Metr, the new product to improve the delivery/conception rate and therefore increasingly improve the farm productivity but preserving the animal welfare, then a shared ethics.
This is our award, with the wish that this certificate, which is today a formal award, tomorrow will become an official acknowledgment, a certification that recognizes the value of your product for which you put so much effort, and that becomes really expendable towards the transformation industry that collects your milk.
AWARDS SafeMilk
SafeMilk Protocols:
a safe and sustainable choice

Higher profit

Better quality

Less waste

Start now!
Request information to our customer service department
or find a retailer close to you
The use of a product like Ozolea-Mast allows you to anticipate the disease outbreak, and decide to intervene even when there are no evident symptoms, with no risks and with no loss of milk: in the last 24 months we reduced the use of antibiotics by 60%, both working with Ozolea-Mast and with a protocol of rapid test or antibiotic sensivity test on the bacterial agent.
Marco Vanzetti
Italian farmer, Graduated in Animal Science Production
The farmer’s profit depends on how quickly he decides to intervene: if he decides to intervene quicky he will have less milk waste and the cow will not be severely affected; if he waits for the disease outbreak, he will have to pay a higher price for the medicine, he will have to withdraw the milk, and the cow will be more affected by the medical treatment, causing an overall reduction in productivity.
Paola Gandolfi
Italian farmer and DVM
O Z O L E A
For you,
for animals,
for the environment.
Home
OZOLEA-MAST
OZOLEA-METR
Protocolli
Testimonianze
Ozolea
Scientific area
Retailers and contacts
Media